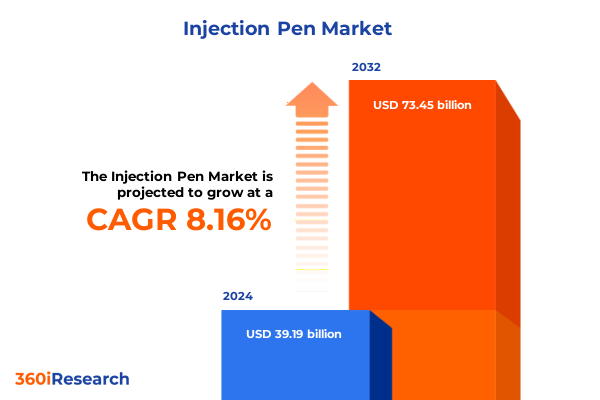
The Injection Pen Market size was estimated at USD 42.03 billion in 2025 and expected to reach USD 45.08 billion in 2026, at a CAGR of 8.29% to reach USD 73.45 billion by 2032.
Setting the Stage for the Future of Injection Pens in Evolving Healthcare Environments Driven by Innovation and Patient-Centric Solutions
Injection pens have revolutionized the administration of injectable therapies, delivering enhanced precision, convenience, and adherence for patients managing chronic conditions such as diabetes, growth disorders, autoimmune diseases, and multiple sclerosis. By minimizing dosing errors and simplifying the injection process, these devices have become an integral component of patient-centric care strategies. As health systems worldwide place greater emphasis on improved outcomes and reduced treatment burdens, the adoption of pen injectors has accelerated, shifting the paradigm of self-administration away from traditional vial-and-syringe methods.
Over the past decade, manufacturers have responded to evolving patient and provider demands by integrating features such as dose memory, auto-lock safety mechanisms, and ergonomic designs. Concurrently, regulators have introduced guidance to ensure device reliability and user safety, further bolstering confidence in pen-based delivery solutions. This convergence of technological innovation and regulatory support has catalyzed investment from both established pharmaceutical companies and emerging device specialists, fueling robust competition and driving continuous enhancements in performance and usability.
Looking ahead, the introduction of digital connectivity and data-driven adherence programs promises to redefine patient engagement and therapy optimization. With the landscape poised for rapid transformation, stakeholders must stay informed about the emerging trends that will shape market trajectories, investment priorities, and care outcomes in the injection pen sector.
Uncovering Transformative Shifts Reshaping the Injection Pen Landscape from Technological Breakthroughs to Value-Based Care Redirecting Growth Paths
The injection pen landscape is undergoing a profound evolution propelled by breakthroughs in smart technologies, integrated ecosystems, and value-based care initiatives. Digital pens equipped with Bluetooth connectivity now enable real-time transmission of dosing information to mobile applications, creating opportunities for remote monitoring and personalized treatment plans. These connected solutions are further supported by artificial intelligence-driven analytics that can identify adherence patterns, predict risk factors, and recommend timely interventions to healthcare providers.
Simultaneously, the rise of value-based care models has shifted the focus from volume to outcomes, incentivizing providers and payers to partner with device suppliers capable of demonstrating measurable improvements in patient health. Pharmaceutical companies have begun collaborating with technology firms to co-develop platforms that integrate injection pens with telehealth services, virtual coaching, and digital therapeutics, ensuring a seamless patient journey from prescription to administration.
Additionally, the convergence of 3D printing, advanced polymers, and human-factors engineering is enabling bespoke pen designs tailored to specific patient populations. Adaptive dosing interfaces for pediatric and elderly users, along with voice-guided instructions, exemplify the ongoing push toward inclusivity and accessibility. As industry leaders prioritize interoperability, data security, and user experience, the injection pen market is set to witness transformative shifts in how therapies are delivered, monitored, and optimized.
Examining the Cumulative Impact of United States Tariffs Implemented in 2025 on Material Sourcing Production Costs and Supply Chain Resilience
In early 2025, the United States implemented a new tranche of tariffs on raw materials and components critical to injection pen manufacturing, including medical-grade plastics, aluminum alloys, and specialized electronics. These measures were introduced as part of broader trade negotiations aimed at reinforcing domestic production but have resulted in escalated procurement costs for device makers. The immediate effect has been a rise in landed costs, prompting companies to reassess supplier portfolios and negotiate new contracts to mitigate margin compression.
To accommodate the increased expenses, some manufacturers have accelerated efforts to localize production, investing in North American facilities to reduce exposure to import duties and streamline supply chains. However, the shift has also revealed capacity constraints and raised questions about the availability of skilled labor for high-precision manufacturing. As a countermeasure, organizations are exploring partnerships with contract manufacturers that possess established in-region operations and quality systems certified to meet FDA and ISO standards.
Moreover, the tariffs have underscored the need for resilient sourcing strategies, spurring investments in alternative materials and multi-tier supplier networks. Companies are diversifying component procurement across multiple geographies, incorporating near-shoring and dual-sourcing approaches to ensure continuity. While these adjustments entail upfront capital expenditures and logistical complexity, they are expected to fortify the injection pen ecosystem against future trade volatility and enhance supply chain resilience.
Revealing Key Segmentation Insights Illuminating Therapy Area Product Type End User and Distribution Channel Drivers for Injection Pen Adoption
Segmentation analysis of the injection pen market reveals nuanced insights driven by therapy area, product type, end user, and distribution channel. Within therapy areas, insulin applications dominate, with rapid-acting, long-acting, intermediate-acting, and premix formulations each exhibiting distinct usage patterns influenced by patient demographics and provider preferences. Growth hormone treatments maintain steady demand among pediatric and adult populations, while advances in biologic therapies for multiple sclerosis and rheumatoid arthritis have spurred interest in targeted self-injection devices tailored to immunomodulatory regimens.
From a product-type perspective, prefilled pens continue to attract significant adoption due to their convenience, ease of use, and reduced contamination risk, whereas reusable systems appeal to cost-sensitive markets and sustainability-focused segments. Disposable pens, characterized by single-use simplicity, offer an alternative for patients with limited storage requirements or short-term treatment plans. The choice among these formats hinges on therapy duration, healthcare reimbursement policies, and patient preferences for convenience versus environmental considerations.
End users such as clinics and hospitals rely on pen injectors to streamline in-office administrations and reduce preparation time, while home care settings increasingly favor pens that integrate connectivity and dosage tracking. Retail pharmacies have emerged as crucial touchpoints, educating patients on proper device usage and facilitating immediate pen dispensation. In distribution channels, hospital pharmacies remain a core conduit for complex therapies requiring clinical oversight, online pharmacies provide discreet, direct-to-patient access for chronic treatments, and retail pharmacies offer familiar, accessible distribution networks that support patient counseling and adherence initiatives.
This comprehensive research report categorizes the Injection Pen market into clearly defined segments, providing a detailed analysis of emerging trends and precise revenue forecasts to support strategic decision-making.
- Product Type
- Therapy Area
- End User
- Distribution Channel
Unearthing Regional Dynamics Highlighting Growth Opportunities Challenges and Ecosystem Collaboration across Americas Europe Middle East Africa and Asia-Pacific
Regional dynamics in the injection pen market underscore how demographic trends, regulatory environments, and healthcare infrastructure shape industry trajectories. In the Americas, robust reimbursement frameworks and high prevalence of diabetes and autoimmune disorders drive sustained demand. The United States, in particular, exhibits a sophisticated ecosystem of device innovation, supported by venture-backed startups and established pharmaceutical giants, while Canada emphasizes accessibility through public health programs and formulary negotiations.
Within Europe, the Middle East, and Africa, diverse healthcare systems present both opportunities and challenges. Western European nations benefit from centralized tendering processes and stringent device standards, leading to rapid uptake of advanced pen solutions. Meanwhile, emerging markets in the Middle East and Africa face infrastructure hurdles but display significant growth potential as governments prioritize chronic disease management. Manufacturers are tailoring their strategies with differentiated pricing models, local collaborations, and training programs to penetrate these heterogenous landscapes.
The Asia-Pacific region demonstrates the fastest adoption curves, fueled by large populations, rising healthcare expenditure, and government initiatives to enhance self-care capabilities. Japan’s aging demographic has prompted widespread integration of user-friendly pens, whereas China’s domestic market is bolstered by supportive policies for biosimilars and digital health platforms. In Southeast Asia, increasing awareness of chronic disease prevention and management is driving diverse stakeholders-from public-private partnerships to local distributors-to invest in education, distribution, and after-sales support models.
This comprehensive research report examines key regions that drive the evolution of the Injection Pen market, offering deep insights into regional trends, growth factors, and industry developments that are influencing market performance.
- Americas
- Europe, Middle East & Africa
- Asia-Pacific
Identifying Leading Companies Shaping the Injection Pen Market through Strategic Partnerships Valuable Patent Portfolios and Operational Excellence
Leading companies in the injection pen sector are distinguishing themselves through strategic partnerships, robust patent portfolios, and relentless focus on operational excellence. Global pharmaceutical leaders continue to leverage their therapeutic expertise, driving incremental enhancements to pen designs while securing regulatory approvals for next-generation platforms. Collaborations with technology providers have become commonplace, enabling seamless integration of sensors, connectivity modules, and data-analytics solutions that enhance patient adherence and clinical oversight.
Specialized device manufacturers are also making significant strides. By securing broad patents for mechanism designs, dosing accuracy, and safety features, these firms are creating high barriers to entry and protecting their innovations in key markets. Many have established joint ventures with contract development organizations to expedite product development and scale manufacturing capacity. This collaborative approach has reduced time-to-market for complex combinational products and enabled manufacturers to meet evolving regulatory requirements efficiently.
In parallel, several agile players have invested heavily in digital ecosystems, offering subscription-based adherence programs and remote monitoring services. These offerings not only generate recurring revenue streams but also provide unique data insights that inform future product roadmaps. As competition intensifies, companies that maintain a balanced portfolio of patented technologies, digital capabilities, and manufacturing agility will be best positioned to capture emerging demand and shape the future of the injection pen domain.
This comprehensive research report delivers an in-depth overview of the principal market players in the Injection Pen market, evaluating their market share, strategic initiatives, and competitive positioning to illuminate the factors shaping the competitive landscape.
- Antares Pharma, Inc.
- B. Braun SE
- Becton Dickinson and Company
- Biocon Limited
- Boehringer Ingelheim International GmbH
- Eli Lilly and Company
- Enable Injections Ltd.
- F. Hoffmann-La Roche Ltd.
- Gerresheimer AG
- Johnson & Johnson
- Lupin Limited
- Medtronic plc
- Nemera SA
- Novartis AG
- Novo Nordisk A/S
- Owen Mumford Ltd
- Pfizer Inc.
- Recipharm AB
- Sandoz Inc.
- Sanofi S.A.
- SHL Group Ltd.
- Stevanato Group SpA
- Ypsomed Holding AG
Actionable Recommendations Empowering Industry Leaders to Innovate Enhance Patient Engagement and Strengthen Supply Chains in the Injection Pen Ecosystem
To capitalize on the evolving injection pen landscape, industry leaders should prioritize the development of connected devices that seamlessly integrate with telehealth and remote patient management platforms. By embedding advanced sensors and leveraging secure data transmission protocols, companies can offer differentiated value propositions focused on adherence monitoring and personalized dosing recommendations. Such innovations not only enhance clinical outcomes but also create new service-based revenue models.
In addition, diversifying supply chains is critical to mitigate the impact of trade disruptions and raw material tariffs. Implementing dual-sourcing strategies, investing in near-shoring initiatives, and collaborating with experienced contract manufacturers will strengthen operational resilience. Concurrently, engaging early with regulatory agencies to align on novel device features and digital health components can expedite approvals and reduce time-to-market.
Finally, companies should deepen partnerships with payers and healthcare providers by demonstrating the economic and clinical benefits of injection pen solutions. Generating real-world evidence through pilot programs and observational studies will bolster reimbursement negotiations and support value-based contracting. By aligning product development, supply chain management, and evidence generation efforts, stakeholders can secure a competitive edge and drive sustained growth in the injection pen ecosystem.
Detailing a Robust Research Methodology Integrating Primary Stakeholder Interviews Advanced Data Analytics and Comprehensive Validation Protocols
This research study employed a comprehensive methodology that integrates both primary and secondary data sources to ensure robustness and relevance. Primary research comprised in-depth interviews with key opinion leaders, including endocrinologists, rheumatologists, and procurement specialists across hospital, clinic, and home-care settings. These discussions provided firsthand insights into device performance, user experience, and emerging clinical needs.
Secondary research encompassed rigorous analysis of peer-reviewed journals, industry white papers, regulatory filings, and patent databases. Financial reports and company press releases were examined to track strategic initiatives, partnership announcements, and investment trends. Advanced data analytics techniques, such as trend analysis and cross-segmentation modeling, were applied to quantify thematic patterns and validate qualitative findings.
To further enhance reliability, the study followed a triangulation approach by cross-referencing multiple data streams and conducting validation workshops with an advisory panel of industry veterans. The combined qualitative and quantitative framework ensured that the insights presented reflect real-world dynamics, regulatory landscapes, and technological advancements. This layered methodology provides stakeholders with a solid foundation for strategic decision-making.
This section provides a structured overview of the report, outlining key chapters and topics covered for easy reference in our Injection Pen market comprehensive research report.
- Preface
- Research Methodology
- Executive Summary
- Market Overview
- Market Insights
- Cumulative Impact of United States Tariffs 2025
- Cumulative Impact of Artificial Intelligence 2025
- Injection Pen Market, by Product Type
- Injection Pen Market, by Therapy Area
- Injection Pen Market, by End User
- Injection Pen Market, by Distribution Channel
- Injection Pen Market, by Region
- Injection Pen Market, by Group
- Injection Pen Market, by Country
- United States Injection Pen Market
- China Injection Pen Market
- Competitive Landscape
- List of Figures [Total: 16]
- List of Tables [Total: 954 ]
Summarizing Critical Findings Emphasizing Strategic Implications and Future-Oriented Perspectives for Stakeholders in the Injection Pen Market
The injection pen market stands at a pivotal juncture where technological innovation, patient engagement, and supply chain resilience converge to redefine how injectable therapies are administered. The transformative shifts toward connected devices and value-based care have created fertile ground for new entrants and incumbents alike to differentiate through digital health integrations, enhanced safety features, and specialized dosing mechanisms.
Tariff-induced cost pressures underscore the importance of diversified sourcing and localized manufacturing strategies, while segmentation nuances across therapy areas, product types, end users, and distribution channels offer granular insights into where targeted investments will yield the greatest returns. Regional variations further demonstrate that tailored market approaches-reflecting reimbursement frameworks, regulatory environments, and demographic profiles-are essential to capture growth opportunities in the Americas, Europe, Middle East, Africa, and Asia-Pacific.
Key players continue to drive competitive intensity through patent filings, strategic alliances, and operational scale-ups. However, those who embrace customer-centric ecosystems-prioritizing data-driven adherence programs, remote monitoring services, and collaborative value propositions-will lead the industry forward. By integrating these critical findings and strategic implications, stakeholders can chart a future-oriented path that aligns innovation with patient needs and business objectives.
Mobilize Your Organization to Capitalize on Injection Pen Innovations with Personalized Support from Ketan Rohom Associate Director of Sales and Marketing
To secure a comprehensive understanding of the injection pen landscape and obtain expert guidance tailored to your organization’s objectives, reach out to Ketan Rohom, Associate Director of Sales & Marketing. Ketan brings extensive experience in guiding stakeholders through complex market dynamics and can help you identify the right scope, deliverables, and customization options to align the research with your strategic imperatives. Engaging with him will allow you to discuss bespoke data requirements, exclusive insights, and priority access to the full report.
By connecting with Ketan, you will gain a direct line to an industry specialist who understands how to translate nuanced research findings into actionable business strategies. He will facilitate prompt delivery of the report, offer additional analysis such as custom deep-dives or executive workshops, and ensure you derive maximum value from your investment. Don’t leave critical decisions to chance-partner with a proven professional whose objective is to empower your team with clarity and confidence.
Contact Ketan Rohom today to secure your copy of the injection pen market research report and embark on a journey toward informed decision-making and sustained competitive advantage.

- How big is the Injection Pen Market?
- What is the Injection Pen Market growth?
- When do I get the report?
- In what format does this report get delivered to me?
- How long has 360iResearch been around?
- What if I have a question about your reports?
- Can I share this report with my team?
- Can I use your research in my presentation?




